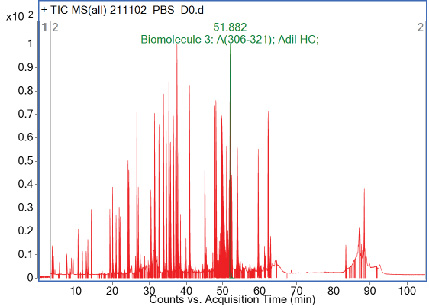

Biophysical characterization of biologics is critical in the development of biopharmaceuticals such as innovator biologics and biosimilars. Many aspects of biologic drug function, activity, and stability are affected by the physical and biological behavior of proteins. Biophysical analytical techniques, for example, can aid in monitoring or confirming conformational integrity, the nature of the protein’s folded state, and how peptides interact to form complex physical structures. Consideration of specific interactions between drug substance and excipients that might potentially alter protein structure and product stability, is critical during formulation development, stability studies, and comparability. Protein degradation and aggregation can also be studied using biophysical techniques.
Aragen has established an advanced state-of-the-art, fully equipped analytical laboratory with highly qualified analytical scientists to perform a comprehensive characterization of biologics. The Analytical lab is equipped with state-of-the-art equipment like Octet/BLI, Biacore/SPR, UV-Vis, SCIEX/PA800 Plus, LC-MS/Q-ToF, Wyatt/MALS, UNCLE/SLS/DLS, and SpectraMax multimode plate readers to perform high-throughput analytics. We have the capabilities to characterize the biologics from clients and those produced at Aragen. All biologics analyzed undergo stringent QC checks before shipping to the client.










