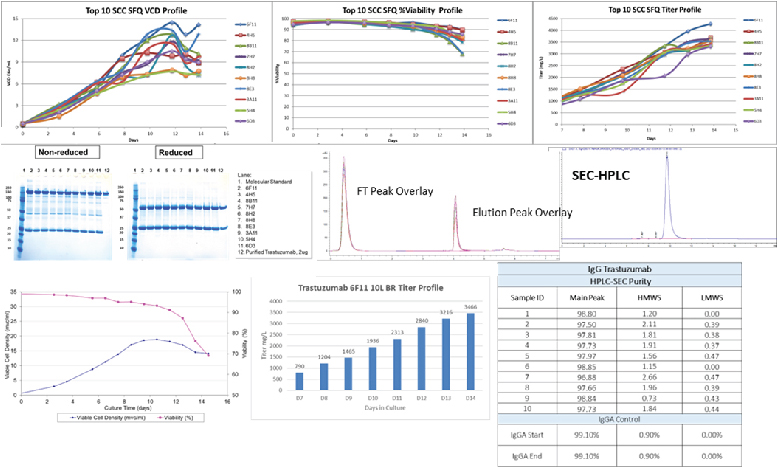
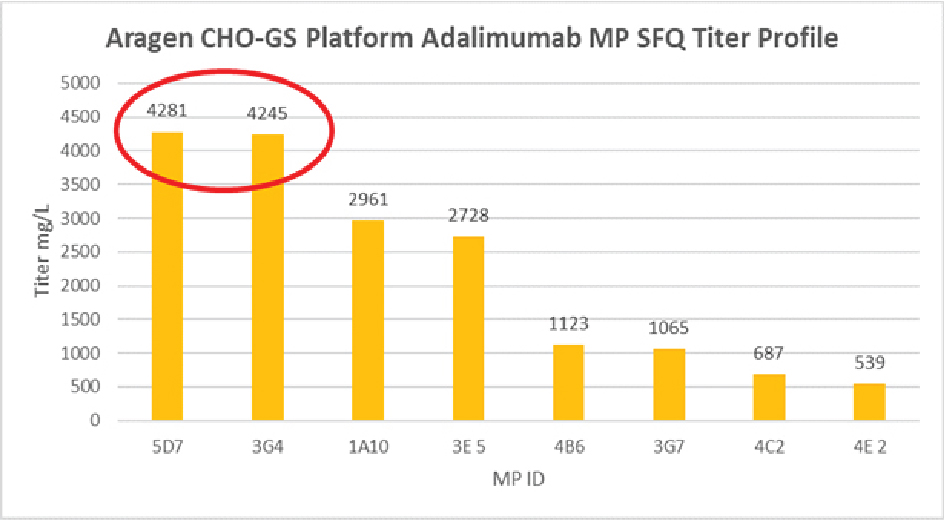
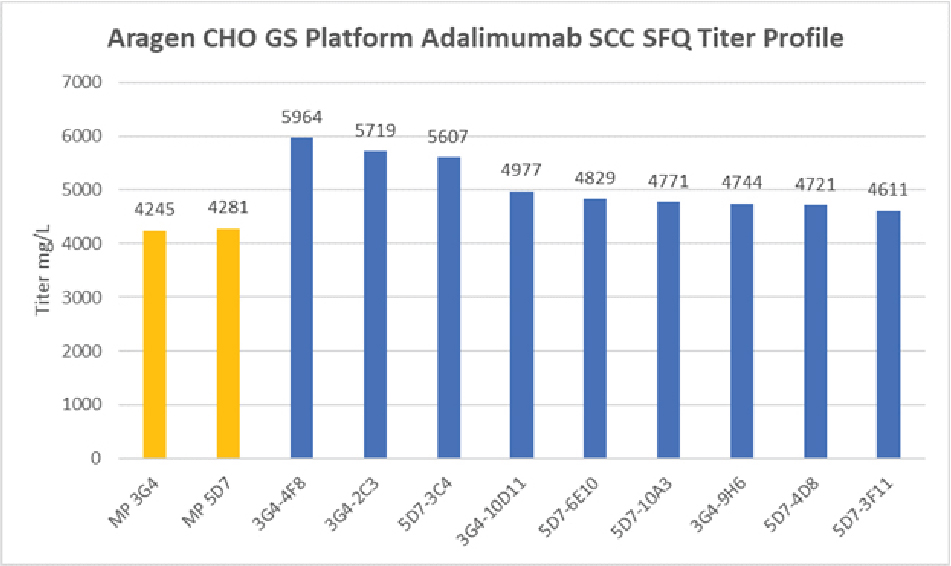
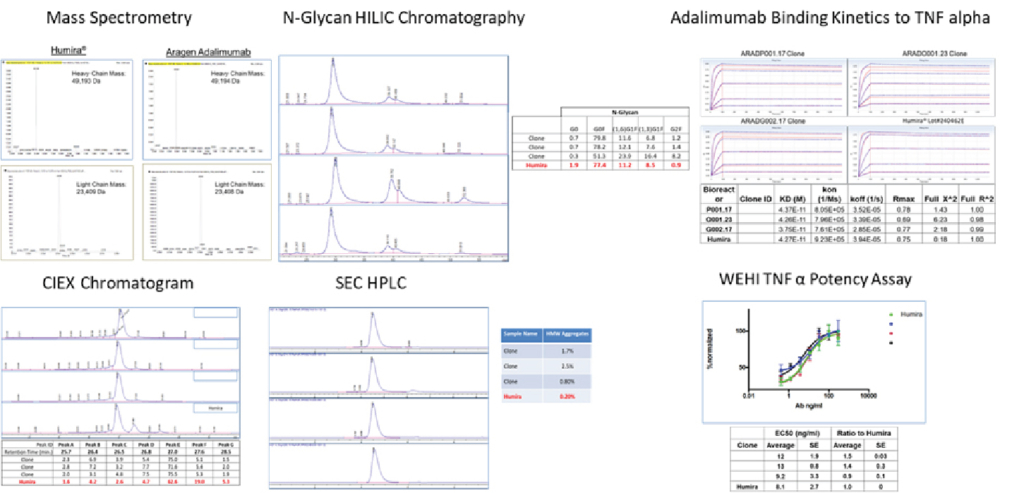
Over the past decade, the Aragen team has successfully expressed biologics including antibodies, antibody fragments, bispecific antibodies, fusion proteins, hormones, and growth factors. Aragen offers high-expressing cell lines for Biosimilar
development.