
Calculating the percentage average efficiency of four commercially available transfection reagents (Reagent 1, Reagent 2, Reagent 3, Reagent 4) in transfecting the mammalian expressing plasmid containing GFP (Green Fluorescent Protein expressing) gene in HEK293 cells and further calculating the percent average viability of the cells
There are three groups of chemical transfection reagents – cationic chemicals like calcium phosphate, cationic lipids, and cationic polymers (Kim and Eberwine, 2010). Cationic chemicals have been found to be cytotoxic and inefficient in majority of situations. The most widely used transfection reagents are Lipofectamine 2000, Lipofectamine 3000, Xtereme Gene HP and polyethylenimine (cationic branched polymer). Lipofectamine 2000 consists of a mixture of 2′-(1″,2″-dioleoyloxypropyldimethyl-ammonium bromide)-N-ethyl-6-amidospermine tetratrifluoroacetic acid salt (DOSPA) and 1,2-dioleoyl-sn-glycero-3-phosphatidylethanolamine (DOPE). It complexes with DNA to allow them to overcome the electrostatic repulsion of the cell membrane. In Lipofectamine 3000, cationic lipid molecules (DOSPA and DOPE) are formulated with a neutral/helper co-lipid (P3000 reagent). Such formulation increases transfection efficiency of nucleotide molecules in both dividing and non-dividing cells under in vitro conditions. Cationic polymers are gene delivery vehicles with high solubility in aqueous solution. DNA polyplexed with cationic polymers like PEI is prevented against degradation by cellular nucleases and induce the endosomal escape. Cationic polymers present ability to more efficient DNA condensation in comparison to cationic lipids.
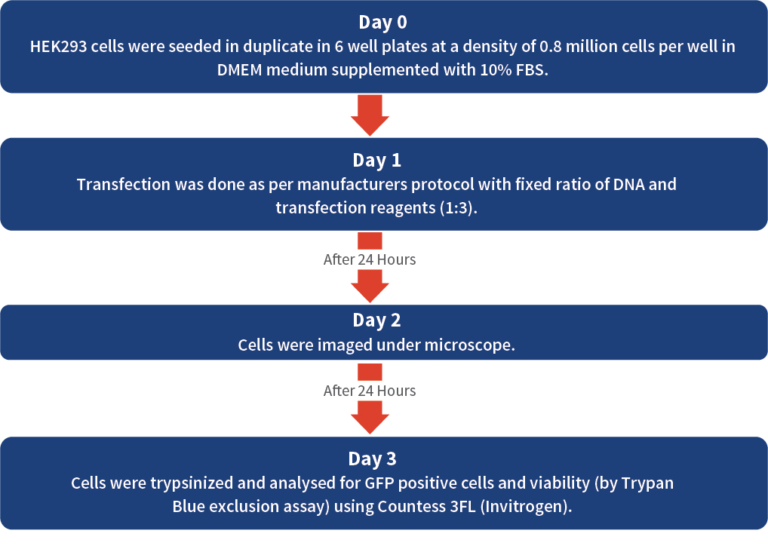
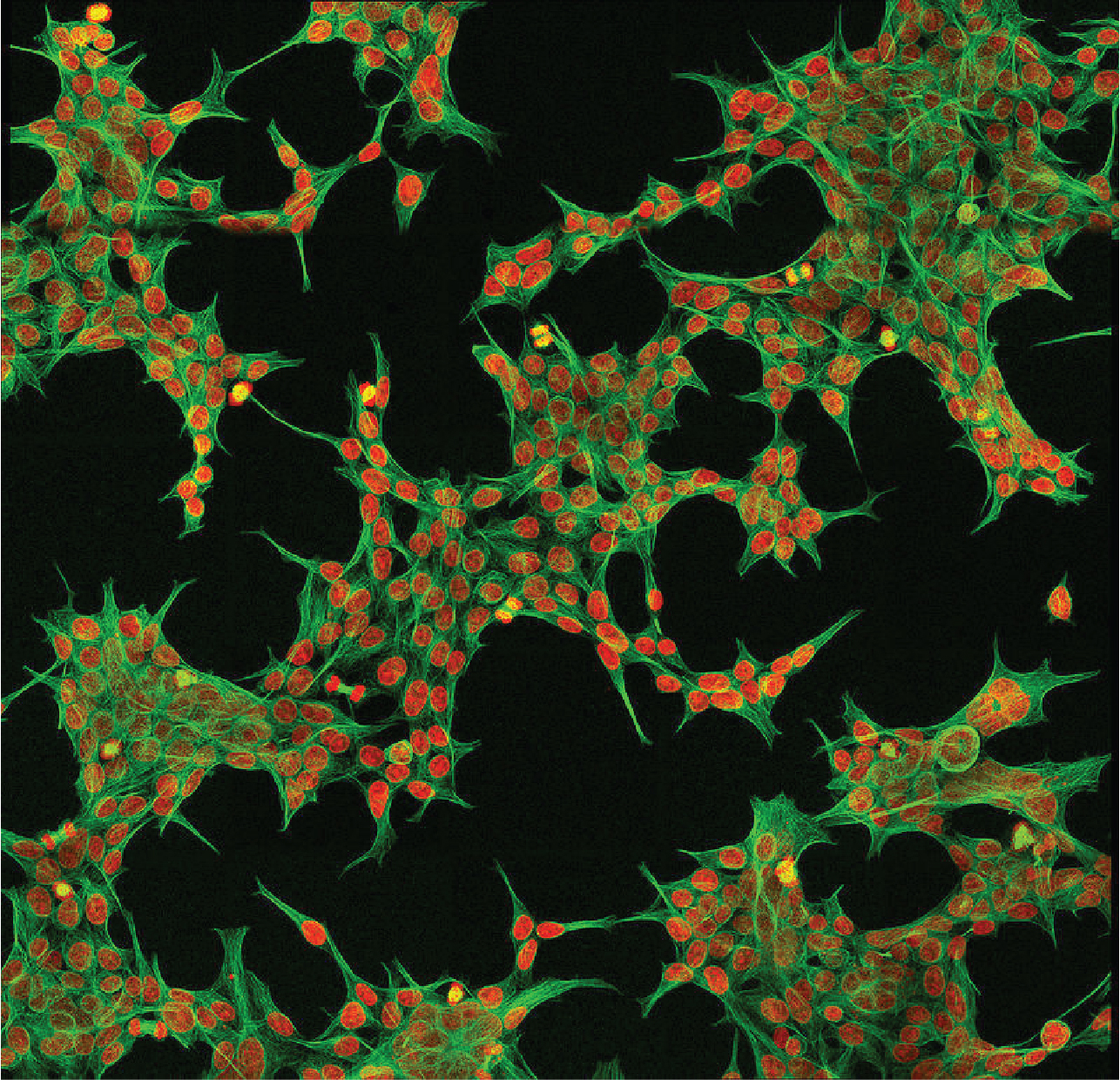
Mammalian expressing plasmid DNA harboring GFP was used for easy identification under fluorescence microscope. For representation of data, arithmetic mean (average) was calculated for the e iciency and viability data among the duplicates. More details in Table 1.
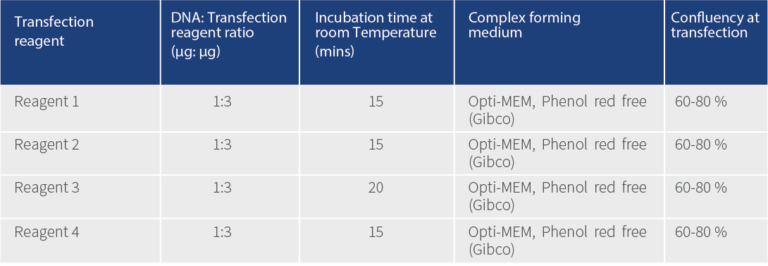
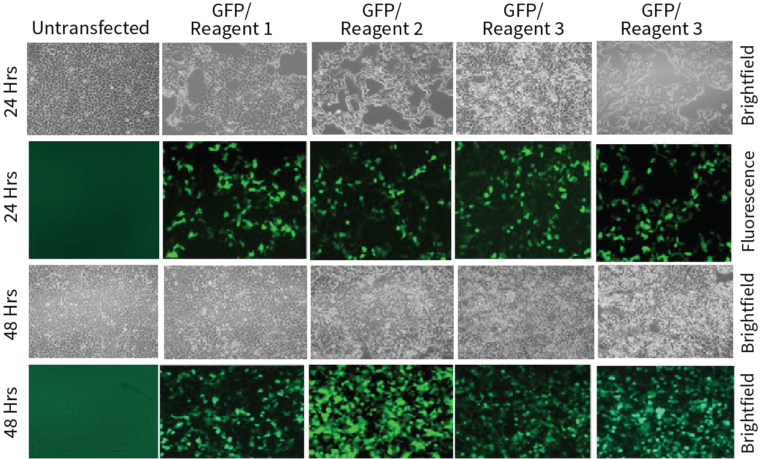
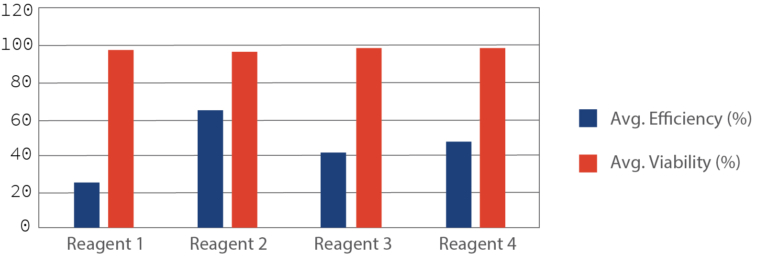
Transfection Reagent 2 is ~3 fold more efficient than Reagent 1 and ~1.5 fold more than Reagent 2 and Reagent 4 (Figure 2). All the reagents showed similar efficiency after 24 hrs of transfection under fluorescence microscope (Figure 1). All the transfection reagents showed comparable cell death after 48 hours of transfection (Figure 2). In brief, Transfection Reagent 2 is the best among the ones we tested in terms of efficiency and viability and all the four reagents have similar cytotoxic effects.
In this study, scientists from Aragen’s Discovery Biology (Cell and Protein Sciences) compared the transfection efficiency and viability of several reagents in HEK293 cells. The results showed that Transfection Reagent 2 had the best transfection efficiency with good viability among tested reagents, making it the top choice for transfecting HEK293 cells. The results from the study can have important implications on gene expression and cell line engineering experiments as it helps to identify the most effective transfection reagent and provides a basis for future research in this area. To know more details about these experiments, details for the transfection reagents tested or to speak with our subject experts, write us at bd@aragen.com.
1. Kim, T.K. and Eberwine, J.H., 2010. Mammalian cell transfection: the present and the future. Analytical and bioanalytical chemistry, 397, pp.3173-3178.
