
Poor solubility has become an increasingly critical barrier to oral bioavailability and clinical success, driven by the growing complexity of New Chemical Entities (NCEs). Contract Research, Development, and Manufacturing Organizations (CRDMOs) play a pivotal role in overcoming this challenge by implementing advanced enabling formulation technologies. Among the diverse enabling formulation technologies available today, spray drying, and nano-sizing-based approaches represent the two most widely adopted strategies for improving the solubility and bioavailability of poorly soluble NCEs.
At Aragen Life Sciences, we leverage material science, pharmaceutics, and particle engineering concepts to design scalable, high performance nanosized formulations specifically tailored for poorly soluble NCEs. This white paper outlines the fundamental principles and development strategies underlying nanosizing technologies, while emphasizing their practical translation from the lab to clinic settings.
A significant majority of NCEs in discovery pipelines exhibit poor water solubility, which can prevent even the most promising drug candidates from achieving therapeutic concentrations in vivo. As molecules become more complex, the frequency of poor solubility issues continues to rise, posing substantial challenges during drug development and clinical translation.
To address these hurdles, a range of enabling formulation technologies have been explored. Foremost among them are spray drying—which creates amorphous solid dispersions—and nanosizing-based approaches, such as nanocrystal and nanosuspension formulations. Among these technologies, nanosizing offers a powerful approach by improving dissolution kinetics through particle size reduction, increased surface area, and curvature-induced solubility enhancement. While spray drying enables the creation of amorphous solid dispersions, nanosizing technologies provide a distinct yet complementary pathway by significantly increasing surface area and thereby enhancing local solubility and dissolution rates.
Evidence from peer-reviewed literature and industry surveys indicates that collectively amorphous solid dispersions (ASDs) and nanocrystal formulations constitute the majority of enabling formulation approaches employed for Biopharmaceutics Classification System (BCS) compounds—Class II and IV, advancing through clinical development. Selection between these platforms is typically informed by the specific physicochemical properties of the Active Pharmaceutical Ingredient (API), desired dose loading, formulation stability, and targeted pharmacokinetic profile.
At Aragen Life Sciences, our nanosizing programs are designed with end-to-end integration, encompassing everything from pre-formulation screening to identification of proof-of-concept drug products for clinical evaluation.
The Noyes-Whitney equation describes dissolution as a function of surface area (A), saturation solubility (Cs), and diffusion properties:
![]()
Two critical insights from this equation inform particle size strategies in formulation:
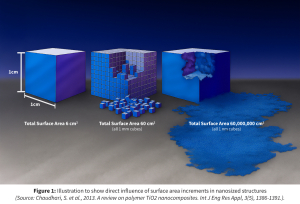
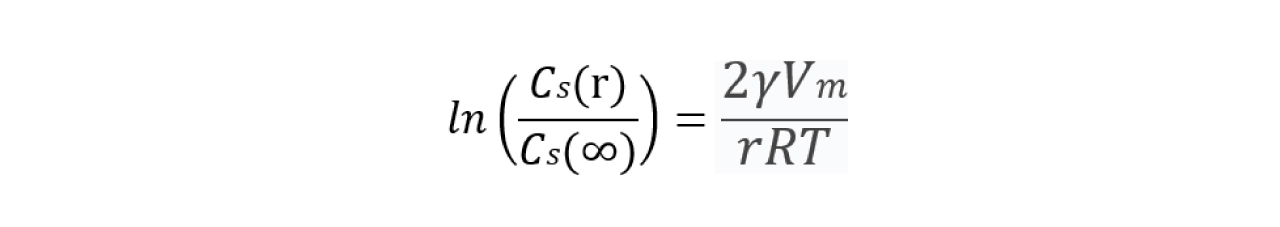
This dual impact—enhancing both surface area and saturation solubility (figure 2) makes nanosizing a powerful enabler in improving dissolution rates, as defined by the Noyes-Whitney equation.
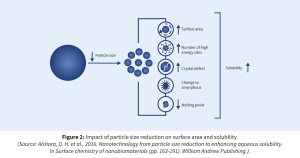
The convergence of geometric (surface area) and thermodynamic (saturation solubility) enhancements expand the possibilities for poorly soluble compounds. This dual impact is pivotal in achieving supersaturation in vivo, producing the well-known “spring and hover” effect, where rapid initial dissolution (spring) is followed by sustained supersaturation (hover) using precipitation inhibitors or stabilizing polymers.
In the field of poorly soluble NCEs, several types of nano-systems are employed to enhance solubility and bioavailability, with nanosuspensions, nanocrystals, and nanoparticles being the most prominent. While these terms are often used interchangeably, they refer to distinct systems with specific characteristics.
Nanocrystals are pure drug particles, typically ranging from 100–500 nm in size, stabilized by surfactants or polymers to prevent aggregation. They retain their crystalline structure and offer enhanced dissolution by increasing surface area and leveraging curvature-induced increases in saturation solubility. Nanocrystals are usually processed further through downstream operations to obtain the final dosage form.
Nanosuspensions are nano-sized drug particles dispersed in water with the aid of surfactants and/or polymers, forming a physically stable colloidal system. They are particularly advantageous for high-dose, poorly soluble drugs—often in preclinical formulations such as oral liquids, concentrated suspensions, or semi-solids forms. If required, nanosuspensions can be converted into powder or solid dosage forms using subsequent downstream processes like top spray or spray drying.
In contrast, nanoparticles generally refer to more complex systems in which the drug is encapsulated or embedded within a polymeric or lipid matrix (e.g., polymeric nanoparticles, solid lipid nanoparticles). These systems not only improve solubility but can also provide controlled release, targeted delivery, or enhanced permeability, depending on their design.
The selection among these nano-systems depends on the specific properties of the drug, the desired release profile, regulatory considerations, and the feasibility of manufacturing for clinical development.
Nanosized drug particles are thermodynamically unstable and prone to aggregation or recrystallization. Effective stabilization is critical for maintaining formulation performance and shelf life. Key stabilizing approaches include:
At Aragen Life Sciences, we perform comprehensive excipient screening and drug-excipient compatibility studies to identify optimal stabilizers that prevent Ostwald ripening, aggregation, and sedimentation during formulation and storage.
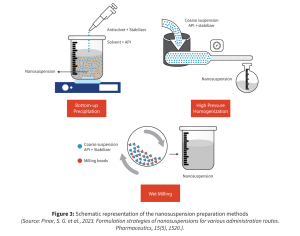
A variety of techniques are available to achieve nanosized NCEs, mostly via top down or bottom-up particle engineering concepts, as depicted in figure 3. Technology selection depends on compound properties, scale, and target dosage form.
Top-Down Approaches
Bottom-Up Approaches
Hybrid and Advanced Methods
For successful development of nanosized drug products, the following parameters are critical:
At Aragen, we employ integrated pre-formulation tools and scale-down batch runs to streamline the pathway to clinical readiness.
Advancing nanosizing approaches from the laboratory to GMP manufacturing requires careful consideration:
Nanotechnology and particle size reduction represent a frontier for enabling the clinical success of poorly soluble molecules. By addressing both thermodynamic (saturation solubility) and kinetic (surface area) barriers, nanosizing strategies offer a robust toolkit for unlocking the full bioavailability potential of NCEs.
At Aragen Life Sciences, we combine scientific rigor with operational excellence to deliver scalable, high-performance nanosized formulations, accelerating drug development, streamlining clinical timelines, and driving commercial success.
We offer end-to-end support throughout formulation design, material characterization, and early-phase manufacturing to enable effective nanosizing development. To further strengthen our capabilities, we are expanding our advanced equipment portfolio
with the addition of high-pressure homogenizers, media mills, and cGMP-compliant suspension handling systems.
