
The development of cancer therapies increasingly demands predictive preclinical efficacy models that reflect the complexity of human tumors. Traditional single modality based preclinical efficacy evaluation in vivo often fail to capture tumor heterogeneity, immune interactions, and pharmacologic nuances, limiting their translational relevance. Integrated in vivo oncology strategies—combining diverse tumor models with pharmacokinetic/pharmacodynamic (PK/PD) analysis, advanced imaging, and biomarker profiling—provide mechanistic insights, improve predictive accuracy, and reduce clinical risk.
Aragen leverages this integrated approach to generate actionable, translational data that guide dosing, patient selection, and clinical strategy, enhancing the likelihood of clinical success.
Cancer therapy development faces growing challenges due to tumor complexity and evolving therapeutic modalities. Traditional in vivo studies relying on oversimplified models often lack predictive accuracy because they insufficiently integrate PK/PD analyses, molecular and cellular assays, and imaging techniques. By integrating complementary tumor models with multimodal pharmacological and biomarker analyses, integrated in vivo oncology approaches generate mechanistic, reproducible data with strong translational relevance.
At Aragen, we offer a broad portfolio of validated xenograft, syngeneic, and humanized models integrated with robust PK/PD frameworks, molecular assays, and advanced imaging to support IND submissions and facilitate clinical translation.
No single preclinical model fully captures human cancer biology. Cell-line xenografts are efficient early tools but lack complexity; patient-derived xenografts retain heterogeneity but require immunodeficient hosts; and syngeneic or humanized models support immune-oncology studies but come with species-specific limitations. Orthotopic and metastatic models provide physiological relevance essential for evaluating therapeutic efficacy.
Integrated approaches combine these models with PK/PD analysis, imaging, molecular biomarkers, and immune profiling to create a multidimensional understanding of drug mechanism, efficacy, and safety. Aragen applies this strategy to transform complex data into actionable insights, minimizing late-stage failures and accelerating clinical translation. The diagram below illustrates how multiple data modalities converge to create a cohesive and predictive view of therapeutic performance across in vivo models (Figure 1).
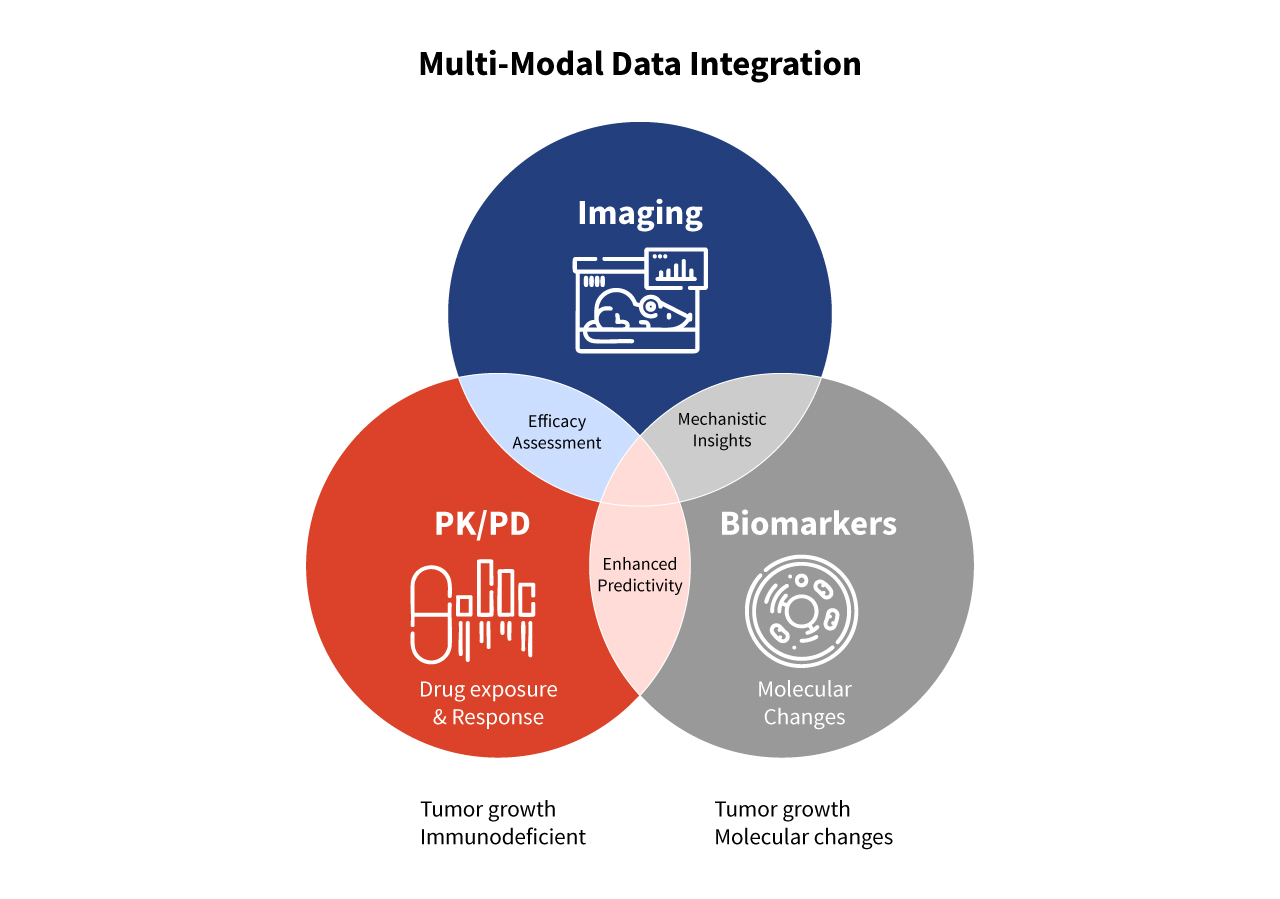
Figure 1: A multi-modal data integration schematic showing how PK/PD, imaging, and biomarker analyses combine to link drug exposure, mechanism, and biological response.
Each oncology program requires a study design tailored to the therapeutic modality, molecular mechanism, and clinical indication. Integrated workflows typically begin with cell-derived xenograft (CDX) studies for rapid screening and initial dose–response assessment. Promising candidates advance to patient-derived xenograft (PDX) models to evaluate heterogeneity and early biomarker signals.
Immuno-oncology agents necessitate syngeneic or humanized mouse models to comprehensively investigate tumor-immune crosstalk, cytokine milieu, and immunomodulatory effects. As development advances, orthotopic and metastatic tumor models assess therapeutic behavior in tissue-specific microenvironments, enabling mechanistic evaluation of metastasis and organ-specific resistance.
Throughout, integrated PK/PD measurements, molecular profiling, and advanced imaging techniques generate interlinked datasets that profile drug exposure, target engagement, and downstream biological responses. This multimodal data strategy enables rigorous mechanistic interrogation and translational extrapolation. Table 1 summarizes key in vivo model characteristics and best-use scenarios.
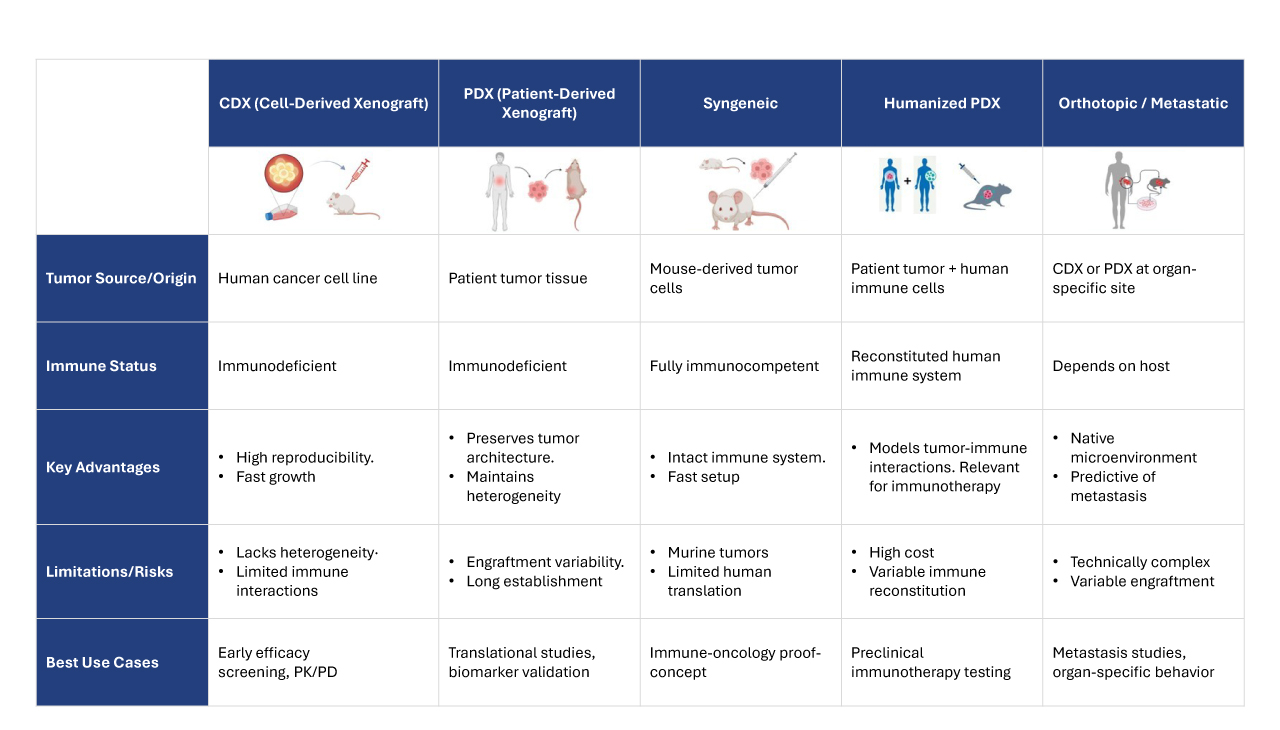
Table 1: Comparative overview of commonly used in vivo oncology models, highlighting their characteristics, translational relevance, and optimal use cases.
Understanding biodistribution, molecular target engagement, and downstream biological effects of a therapeutic is central to predicting its clinical potential. PK/PD integration helps define exposure–response relationships, clarify mechanism, and guide dose selection early in development.
Within this scientific framework, Aragen generates high-resolution exposure data using sensitive LC-MS/MS and immunoassay-based platforms and pairs these measurements with pharmacodynamic markers such as cytokine modulation, pathway activation, and cell-cycle effects.
To enable this integration in practical settings, we employ micro-volume sampling and complementary safety assessments, supporting comprehensive yet low-burden evaluation of pharmacologic activity. Specialized assays further extend this capability to complex therapeutics such as antisense oligonucleotides and aptamers, ensuring robust PK/PD insights across diverse therapeutic platforms.
In vivo imaging provides a temporal and spatial view of tumor biology that cannot be achieved through endpoint measurements alone. Modern optical systems allow continuous, non-invasive tracking of tumor growth, therapeutic distribution, and early biological changes across diverse tumor settings.
Leveraging these capabilities, Aragen employs multimodal platforms—fluorescence, bioluminescence, and X-ray imaging—to monitor treatment responses within the same animals over time. Deep-cooled CCD detection enables sensitive visualization of subtle changes, supporting evaluation of nanoparticle trafficking, biological accumulation, and vector expression while reducing variability and animal use.
Biomarkers play a critical role in connecting preclinical biology to clinical outcomes, offering insights into mechanism, response heterogeneity, and potential patient segmentation. Their integration with PK/PD and efficacy data allows early identification of meaningful pathways and predictive signatures.
Aragen incorporates multiplex biomarker profiling into each study, utilizing Mesoscale discovery (MSD), ELISA and HTRF (Homogeneous Time Resolved Fluorescence) platforms, that allow measuring PD markers with high sensitivity. These datasets are then integrated with exposure and efficacy findings using advanced analytical approaches to reveal mechanistic patterns with strong translational relevance.
Integrated in vivo oncology studies provide a predictive preclinical foundation that reduces clinical risk. Evaluating therapies across diverse biological contexts enables early identification of resistance mechanisms, informs patient-selection strategies, and guides dose optimization before clinical trials. Combining PK/PD, imaging, and biomarker analyses uncovers translational markers for clinical enrolment or companion diagnostics, while assessments across models with varying immune profiles improve prediction of immunotherapy responses. These integrated insights strengthen decision-making and enhance confidence in advancing candidates toward the clinic.
Aragen Life Sciences provides end-to-end customized preclinical efficacy solutions, specializing in oncology, immuno-oncology, and precision therapeutics. By integrating cutting-edge in vivo models, PK/PD analysis, advanced imaging, and biomarker profiling, we deliver mechanistic and translational insights that accelerate drug development.
Transforming preclinical data into actionable insights—unlock predictive insights with Aragen to guide your oncology program.
