
Cancer antigen-expressing cell lines are essential tools for advancing targeted cancer therapies, enabling accurate evaluation of antigen-specific molecules and supporting preclinical discovery workflows. Among key surface markers, B‑cell maturation antigen (BCMA; TNFRSF17/CD269) has emerged as a high‑value therapeutic target due to its selective expression on mature B cells and strong upregulation in multiple myeloma. As BCMA‑directed modalities such as ADCs, bispecific antibodies, and CAR‑T therapies continue to expand, the need for robust, well‑validated BCMA‑expressing cell models has become increasingly critical. This case study highlights Aragen’s development of a stable CHO‑K1 cell line co‑expressing human BCMA and eGFP to support next‑generation immuno‑oncology drug discovery.
To facilitate BCMA-related research, we generated a stable CHO-K1 cell line that co-expresses human BCMA on the cell surface and enhanced green fluorescent protein (eGFP) as a reporter. As outlined in the workflow below, CHO-K1 cells were first engineered to stably express eGFP. A monoclonal population from this line was then used to generate the CHO-K1-eGFP-BCMA cell line.
Stable CHO-K1 cell lines co-expressing eGFP and human BCMA were generated through a sequential reporter line establishment, BCMA integration, and monoclonal isolation.
1. CHO-K1-eGFP Cell Line Development
Flow cytometry confirmed clear eGFP positivity in monoclonal lines derived from the stable pool, including Clone 1 and Clone 2. Both clones showed a pronounced rightward fluorescence shift relative to controls, indicating robust reporter expression with uniform distributions consistent with clonal homogeneity (Figure 1).
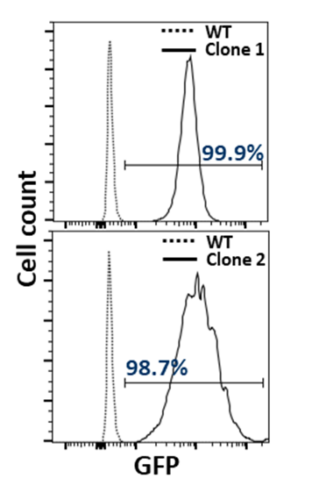
Figure 1: Flow cytometry analysis of CHO-K1-eGFP monoclones.
2. CHO-K1-eGFP-BCMA Stable Pools
CHO‑K1‑eGFP (Clone 1) cells were transduced at an MOI of 2.5 to establish BCMA‑expressing stable cell pool. Evaluation of the engineered stable pools demonstrated surface BCMA expression while retaining eGFP, confirming successful co‑expression on the CHO‑K1‑eGFP background. Majority of events localized to the BCMA⁺/eGFP⁺ gate with compact distributions, indicating efficient enrichment and pool uniformity suitable for downstream cloning (Figure 2).
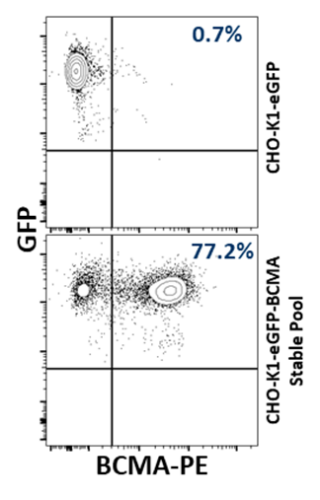
Figure 2: Flow cytometric evaluation of CHO-K1-eGFP-BCMA stable cell pools.
The selected monoclonal line exhibited strong, consistent surface BCMA alongside stable eGFP, confirming the intended double‑positive phenotype at clonal resolution. Tight clustering and narrow fluorescence peaks indicated genetic stability and low intra‑clone variability, establishing the clone as assay‑ready for antibody binding, T‑cell engager evaluation, and anti‑BCMA CAR‑T functional studies (Figure 3).
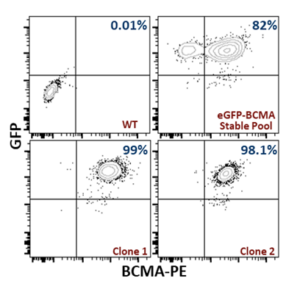
Figure 3: Validation of BCMA and eGFP expression in monoclonal CHO-K1-eGFP-BCMA cell line.
BCMA plays a central role in B‑cell survival and is strongly upregulated in multiple myeloma, making it a key target for emerging immunotherapies, including ADCs, bispecific antibodies, and CAR‑T cells. Reliable BCMA‑expressing cell models are therefore essential for validating antigen‑specific interactions, optimizing engineered therapeutic modalities, and supporting functional potency assays.
Aragen’s CHO‑K1‑eGFP‑BCMA cell line provides a robust, well‑characterized platform for these applications. By co‑expressing surface BCMA and an eGFP reporter, the model enables straightforward detection, assay development, and compatibility with diverse immuno‑oncology workflows—from antibody screening to T‑cell engager and CAR‑T evaluation to support the development of next‑generation BCMA‑targeted therapies.
Aragen delivers reliable Cell & Protein solutions that move programs from concept to preclinical faster.
Partner with Aragen to derisk development and accelerate next-generation oncology therapeutics.
