

Antibody–drug conjugates (ADCs) require multi-analyte quantification to fully characterize pharmacokinetics and drug stability. This study implemented an integrated bioanalytical strategy to quantify total antibody (TAB), intact ADC, and free payload in biological matrices. The workflow combined electrochemiluminescence (ECL) ligand-binding assays and LC–MS/MS to achieve sensitive, selective detection across broad concentration ranges.
Pharmacokinetics were assessed in male (Figure part A) and female (Figure part B) subjects across four doses (3, 10, 30, 60 mg/kg):
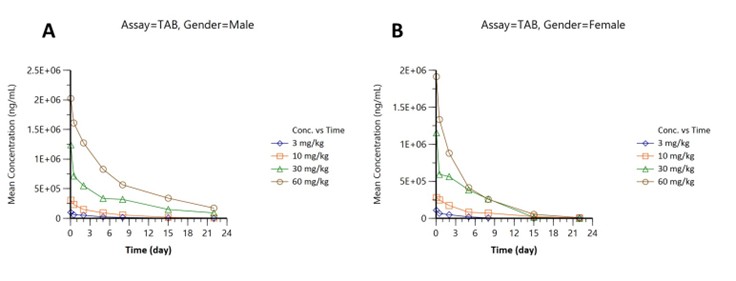
Figure 1: Flow cytometry analysis of CHO-K1-eGFP monoclones.
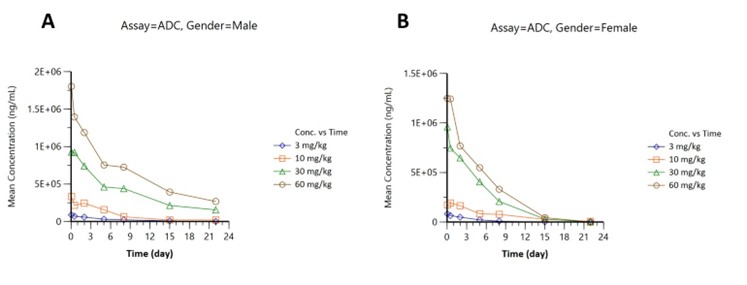
Figure 1: Total antibody (TAB)concentration-time profiles: (A) males, (B) females, doses 3–60 mg/kg.
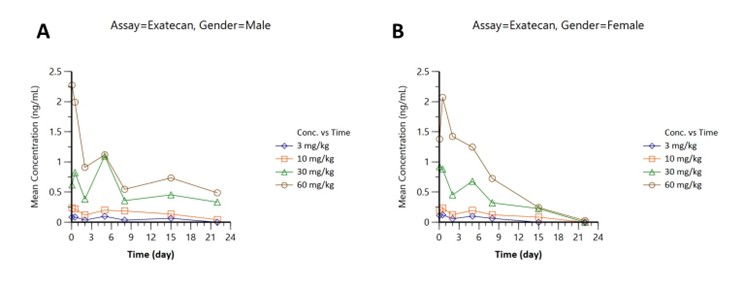
Figure 3: Free payload (Exatecan) concentration-time profiles: (A) males, (B) females, doses 3–60 mg/kg.
These results demonstrate the robustness and sensitivity of the integrated bioanalytical workflow, enabling comprehensive PK characterization across doses and genders.
This case study demonstrates that our integrated ECL and LC–MS/MS workflow delivers fast, sensitive, and reliable quantification of TAB, ADC, and free payload, enabling clients to accelerate ADC development and generate robust pharmacokinetic data.
