

Developing a robust and scalable approach to obtain a high-purity R-isomer is essential for efficient downstream synthesis and regulatory compliance. This need becomes more critical when the existing resolution method suffers from variability, low recovery, and operational complexity, compromising both yield and consistent attainment of specification.
This case study demonstrates how Aragen developed a simplified, data-driven crystallization-based resolution process that improves yield, reduces unit operations, and consistently delivers the required high chiral purity. R-isomer is essential for efficient downstream synthesis and regulatory compliance. This need becomes more critical when the existing resolution method suffers from variability, low recovery, and operational complexity, compromising both yield and consistent attainment of specification.
The initial process relied on forming a diastereomeric salt with dibenzyl tartaric acid (DBTA), breaking the salt, and repeating the salt-formation cycle to reach the required chiral purity. Despite two cycles, the process showed:
We optimized the resolution step to improve recovery, maximize chiral purity, and replace the multistep salt-based workflow with a more predictable crystallization strategy.
The approach combined solubility-based resolution and temperature driven re-crystallization to isolate and refine the R-isomer using optimized acetonitrile/dichloromethane (ACN/DCM) and acetonitrile (ACN)/toluene solvent systems.
1. Solvent Screening for Resolution
A broad solvent screen evaluated the solubility of the free base and R-isomer. ACN/DCM mixtures emerged as the most promising isomer.
Selected system: 20% ACN in DCM for the resolution step, balancing solubility, recovery, and scalability.
Table 1. Low-temperature solubility trends for R- and S-isomers in ACN/DCM (resolution selection).
| ACN % in DCM | Temperature (°C) | R-isomer (mg/mL) | R-isomer (mg/mL) |
| 28% | 5 | 34 | 31 |
| 25% | 5 | 32 | 28 |
| 20% | 5 | 28 | 26 |
| 28% | 15 | 45 | NA |
| 25% | 15 | 37 | NA |
| 20% | 15 | 33.3 | NA |
The data above (Table 1) indicates that reducing acetonitrile content (20%) lowers overall solubility at 5–15 °C, which in turn improves recovery of the R-isomer compared to higher ACN concentrations (e.g., 28% ACN, where recovery was limited to ~25%). isomer compared to higher ACN concentrations (e.g., 28% ACN, where recovery was limited to ~25%). -isomer compared to higher ACN concentrations (e.g., 28% ACN, where recovery was limited to ~25%).
2. Re-Crystallization Solvent System
To enhance the chiral purity beyond the resolution step, ACN/toluene mixtures were explored:
Selected system: 12% ACN in toluene (primary), 16% ACN for poor feedstocks.
Table 2. Temperature-dependent solubility gap for R-isomer in ACN/toluene (recrystallization driver).
| ACN % in Toluene | Temperature (°C) | R-isomer (mg/mL) |
| 12% | 25 | 17 |
| 12% | 60 | 83 |
| 17% | 25 | 49 |
| 20% | 25 | 55 |
At 12% ACN, the R-isomer exhibits a large solubility shift from 17 mg/mL at 25 °C to 83 mg/mL at 60 °C, offering a strong thermal window for controlled crystallization and effective impurity rejection (Table 2).
Key Findings
Table 3. Recrystallization trial summary and outcomes.
| Trial | Batch Size | Input Chiral Purity (R:S) | Solvent System | Final Chiral HPLC (R:S) | Yield (%) |
| 1 | 1 g | 97.7:2.29 | 10% ACN in toluene (12V) | 99.36:0.64 | 65.0 |
| 2 | 5 g | 98:2 | 12% ACN in toluene (12V) | 99.89:0.11 | 66.0 |
| 3 | 20 g | 80:20 | 12% ACN in toluene (12V) | 99.6:0.4 | 60.5 |
| 4 | 34 g | 70:30 | 16% ACN in toluene (12V) | 99.6:0.4 | 44.0 |
Modified Process
The overall optimized process—summarized in the workflow image—replaces the earlier multistep DBTA salt-resolution route with a streamlined, crystallization-based approach. By moving to targeted solvent systems for resolution (20% ACN in DCM) and recrystallization (ACN/toluene), the process eliminates redundant salt-formation/salt-break cycles, reduces batch complexity, and increases overall yield from approximately 18–22% (existing process) to 28–32% (modified process) while consistently meeting the high-purity specification.
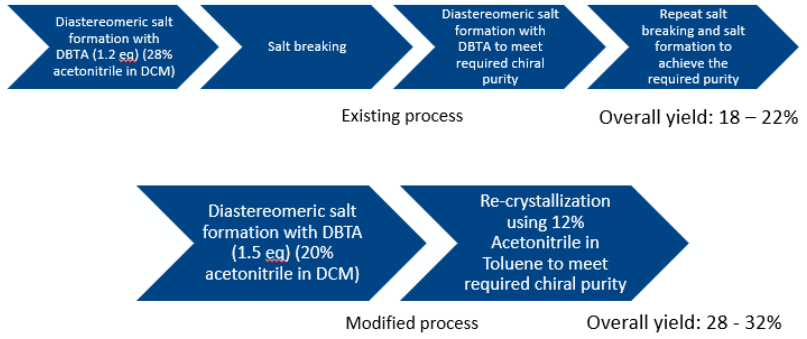
Figure 1: Optimized crystallization-based workflow for R-isomer resolution and purification.
This simplified design ensures reproducible control over purity and recovery, offers a shorter batch cycle time, and demonstrates reliable scalability from gram to multi-tens-of-grams without compromising product quality. -tens-of-grams without compromising product quality.
Using solubility-driven solvent selection and controlled crystallization design, Aragen successfully replaced a yield-limiting, multistep resolution method with a simple, scalable, high-fidelity crystallization process. The optimized workflow ensures consistent high R-isomer purity, improves overall yield (28–32% vs 18–22%), minimizes processing steps, and delivers reproducible performance—ready for seamless scaleup and manufacturing transfer. -driven solvent selection and controlled crystallization design, Aragen successfully replaced a yield-limiting, multi-step resolution method with a simple, scalable, high-fidelity crystallization process. The optimized workflow ensures consistent high R-isomer purity, improves overall yield (28–32% vs 18–22%), minimizes processing steps, and delivers reproducible performance—ready for seamless scale-up and manufacturing transfer.
Aragen brings deep expertise in chiral resolution, crystallization development, and solid-form optimization, enabling clients to overcome challenges related to purity, yield, and scalability. Our integrated capabilities—from solvent and solubility screening to recrystallization design, scaleup, and analytical support—ensure data-driven, robust, and manufacturing-ready processes. With a focus on efficiency, reproducibility, and high-quality deliverables, we help accelerate development of complex intermediates and APIs across diverse chemistries.
